By John “CZ” Czwartacki, Executive Director of the Informed Patients Project
“Informed patients are healthier patients.” For me, like millions of my fellow patients, access to clear, reliable information about treatments isn’t optional. It’s essential. Living with multiple sclerosis for three decades, I have stayed informed in dozens of ways: through caregivers, family, online seminars, peer- reviewed journals, public health news stories, etc. I pursue any and all information that helps me to engage with my doctors – including drug ads.
I have been introduced to new therapies I might have never considered after seeing drug ads. This includes both treatments that my doctors and I rejected and treatments that were prescribed and infused. Drug ads have helped me have informed conversations, improve my life, and fight my illness. Drug ads have helped my friends recognize symptoms better understand their treatment options. Most importantly, these ads give patients information that empowers healthier lives.
This month, U.S. Food and Drug Administration (FDA) Commissioner Dr. Marty Makary made comments on social media about the FDA’s targeting of drug advertising, announcing that the FDA is “just getting started”. Dr. Makary’s statement – and other similar sentiments he has shared in the past – give patients like me pause.
American regulators help ensure that medicines, research, and health information is trusted, verified, and constructive, including its advertising. Today, the existing FDA oversight and enforcement system around drug ads already mandates risk disclosures, monitors promotional materials, and issues warning letters when companies violate the rules. The FDA’s oversight of drug ads is one of the clearest examples of federal oversight doing exactly what it is designed to do.
In fact, for over thirty years, the FDA has ensured drug ads are accurate, balanced, and educational. Companies running drug ads must present side effects clearly. They must avoid misleading claims. They must substantiate what they say. These standards are applied consistently across the entire industry. In 2025 alone, the FDA issued over 200 enforcement letters regarding prescription drug advertisements, underscoring that oversight isn’t theoretical, but ongoing.
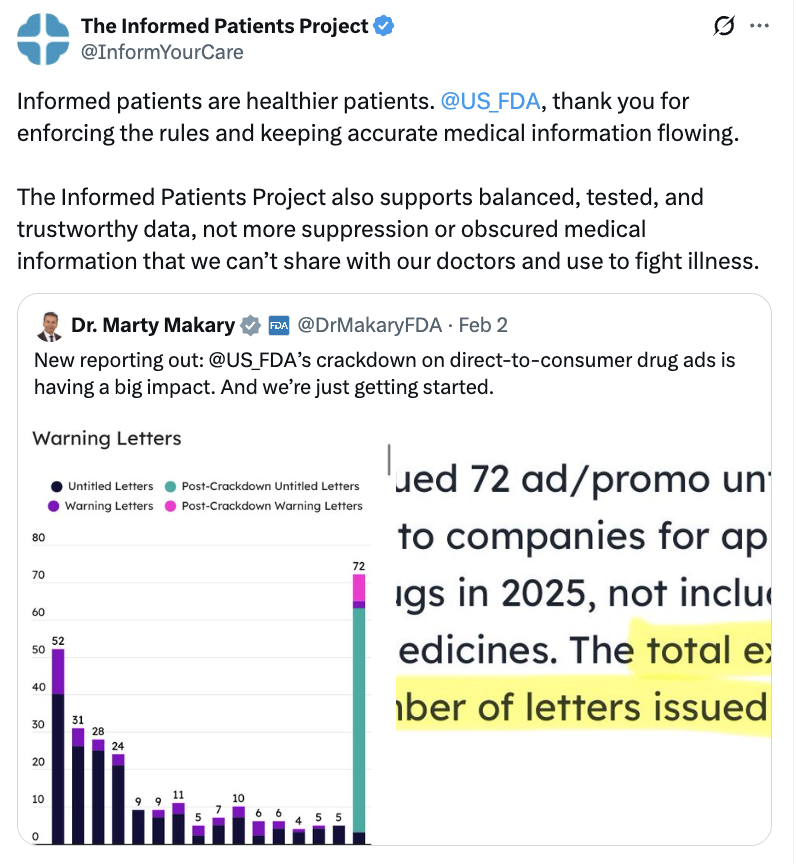
Clear, practical, and uniformly enforced rules help ensure patients get reliable information and stay protected from misleading claims. Under Commissioner Makary’s leadership, enforcement has become more consistent, aligning with the intention behind existing laws. If additional enforcement means patients receive more accurate and useful ads, that’s positive – as long as it remains within the parameter of the current laws and regulations. The goal should always be to improve clarity, increase information and empower patients, not to create unnecessary obstacles.
Strong FDA oversight has encouraged companies to be more careful, more transparent, and more responsible in how they communicate. The mission of ensuring truthful and balanced advertising has been accomplished. That success should be acknowledged.
But when you’re living with a chronic condition, information is not optional. For many patients, drug ads are the first place they hear the name of a new therapy. Drug ads prompt research, conversations, and important questions.
Policymakers often speak about empowering patients and increasing transparency in health care. Preserving the current drug advertising framework is a concrete way to do just that. The FDA has built a system that deters bad actors, enforces standards, and ensures patients receive meaningful information.
It is time to recognize that this framework is achieving its purpose.



